Introduction
Rotator cuff injuries represent a clinical problem, and their repair constitutes a significant surgical challenge. Despite the technical improvements in mechanical repair available to us in 2024, the incidence of tendon re-rupture or failure to heal is very high, reaching 30% according to a recent systematic review involving a follow-up period of over 10 years(1). Patients with repair or healing failures, while showing clinical improvement versus the presurgical condition, have less favourable functional outcomes than those with correct rotator cuff healing(2). In 2024, technological advances in anchoring systems, sutures or the repair technique itself have allowed us to make biomechanically stronger and more stable sutures. However, the re-rupture rate remains about 30%, as commented above, and according to the most recent publications, no clinically significant differences are seen when using different suture methods, whether augmented or not with different types of grafts(1,3,4).
As is well known, chronic rotator cuff tears, if allowed to evolve freely, result in upper displacement of the head of the humerus towards the acromion - with the clinical and biomechanical alterations that this entails(5). In seeking to correct this upper instability, Mihata et al.(6) reported good results in 2012 with superior capsular reconstruction using a fascia lata autograft, aiming to statically stabilize the humeral head and prevent progression towards cuff arthropathy. Other authors have used allografts of the fascia lata, Achilles tendon, ischiotibial muscle, or human dermis patches to reduce morbidity at the autograft donor site(7,8,9,10). Based on this same premise and technique, some authors have used the patient's own long portion of the biceps (LPB), when present at surgery, for superior capsular reconstruction of the glenohumeral joint, leaving the proximal part of the tendon inserted in the upper part of the glenoid and attaching the distal part to the greater tuberosity(9,11,12,13,14). After anchoring in the greater tuberosity, the authors performed a tenotomy/tenodesis of the LPB immediately distal to the insertion of the anchorings in the greater tuberosity. In this way, the biceps tendon simulates the superior capsule, maintaining its original anatomical insertion at the top of the glenoid and at the greater tuberosity with the surgical anchorage completed, thereby correcting the superior displacement generated by the cuff tear.
The biceps tendon is usually exposed at the anterolateral corner defect in supraspinatus tears, and can easily be used to reinforce rotator cuff repair with supplemental sutures. The concept of using autologous LPB as a graft is attractive, because it is locally available and, as such, free of additional costs, theoretically less technically demanding, and potentially faster to perform than augmentation with other heterologous grafts.
Some authors have used this technique in massive irreparable tears as an isolated or adjunctive technique in partial rotator cuff repairs(8,9,11,12,14,15,16).
While Llanos-Rodríguez et al. propose a technique with redirecting of the LPB(16), in the present study we present our version of superior capsular reconstruction with autologous LPB, without deviating the graft from its natural anterosuperior trajectory, as a restriction against upper displacement, as augmentation of partial/total rotator cuff repair, and as a reinforcement and guarantee of such repair. Recent publications show that inclusion of the LPB in the repair procedure improves both the biomechanics and the biology of repair, thereby reducing the re-rupture rate(12).
Presentation of the surgical technique (Video 1)
Indications and contraindications of the technique
- Main indication: this surgical technique is indicated as an adjunct to cuff repair, with the aim of mechanically unloading the repair in the short term and affording biological augmentation in the medium term, as well as restricting anterosuperior humeral head displacement in the long term. It thus requires the presence of repairable cuff tears.
- Relative contraindications: tearing of the cuff that can only be partially repaired according to closure capacity or flayed degeneration of the LPB with delamination of the latter.
- Absolute contraindications: absence of LPB at the time of arthroscopy.
Positioning, preparation and general measures
Regarding patient positioning, arthroscopic portals and initial joint examination, our group performs shoulder arthroscopies under general anesthesia and interscalene plexus block, with the patient in lateral decubitus and the arm in 30-40° abduction with 4-5 kg of traction. A standard or slightly lateralized first posterior portal is established (with subacromial visualization already in mind, allowing a slightly more lateral view over the cuff) and the30° optic is positioned in the glenohumeral joint. In the first intra-articular exploration, supraspinatus lesions are confirmed, the integrity or not of the subscapularis is assessed, the condition of the glenohumeral cartilage and the quality of the LPB tendon are evaluated, and the axillary recess is examined. Regarding the working portals, an anterior portal is made, with access both intra-articular and to the anterior subacromial zone. In turn, an Abbocath No. 16 needle is used to mark the supraspinatus lesion in the position of the lateral portal, and the corresponding working portal is also created. For this portal we use an 8.25 mm × 7 cm Twist-InCannula® (Arthrex, Naples, FL, USA) working cannula, as it will be the portal through which we will work most of the knotting procedures.
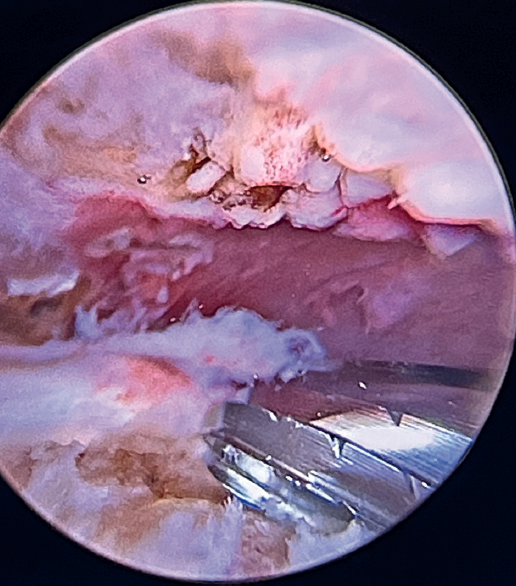
If the patient has an associated subscapularis tendon injury requiring repair, it is at this point that we would perform repair from within the joint.
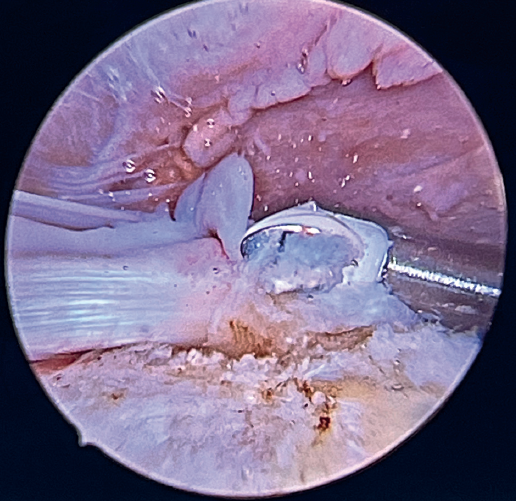
The entry trocar is used to switch from intra-articular to subacromial through the posterior portal. At this point, a wide bursectomy and the freeing of medial adhesions in the tendons is crucial, extending widely towards the spine of the scapula if necessary. In addition, the type of tear, the mobility of the cuff and its reduction to the footprint of the greater tuberosity without tension is assessed by means of grasping forceps.
Preparation of the bone footprint and autologous graft of the long portion of the biceps
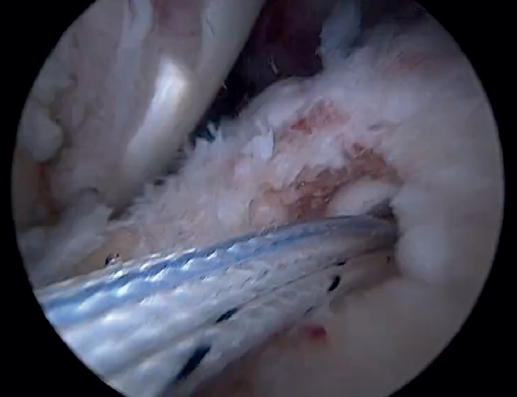
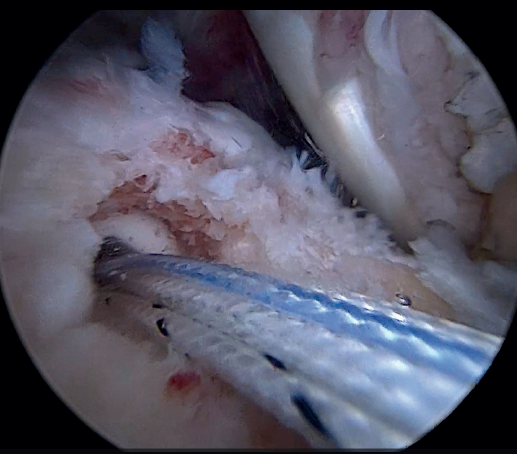
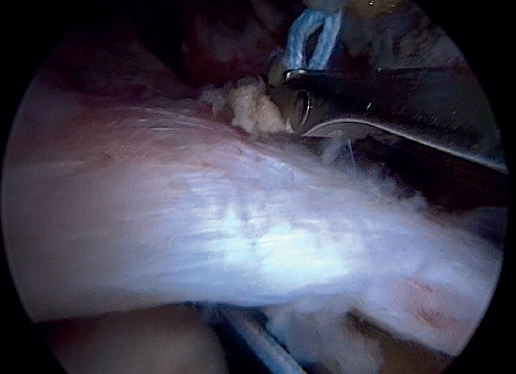
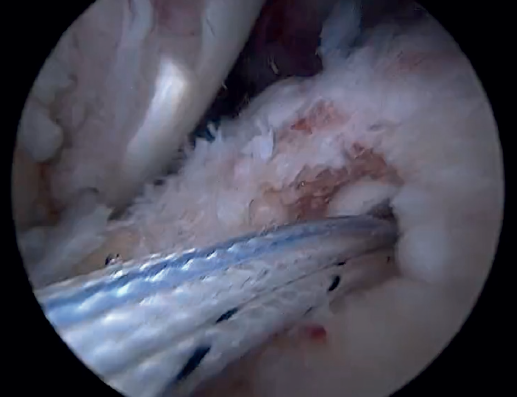
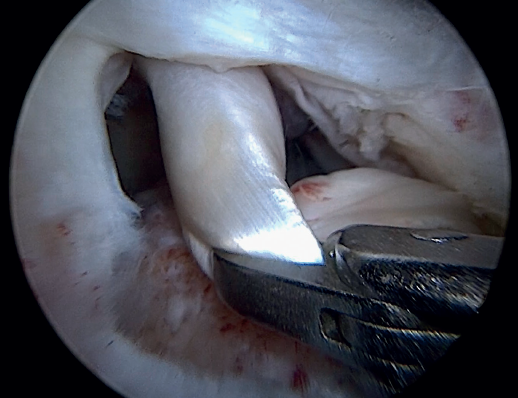
The greater tuberosity is then prepared with the bone drill, creating a bleeding bone surface to enhance tendon-bone healing, as this is where both cuff repair and augmentation with biceps will be carried out (Figure 1).
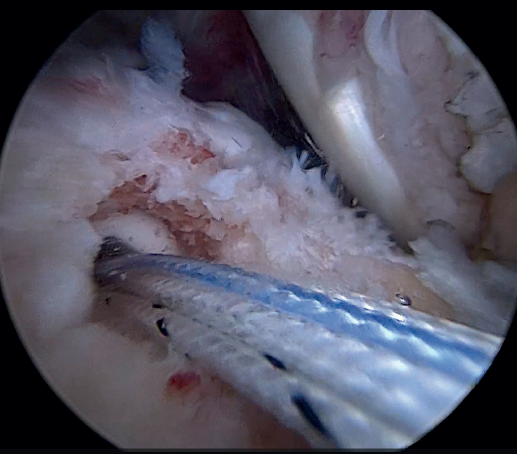
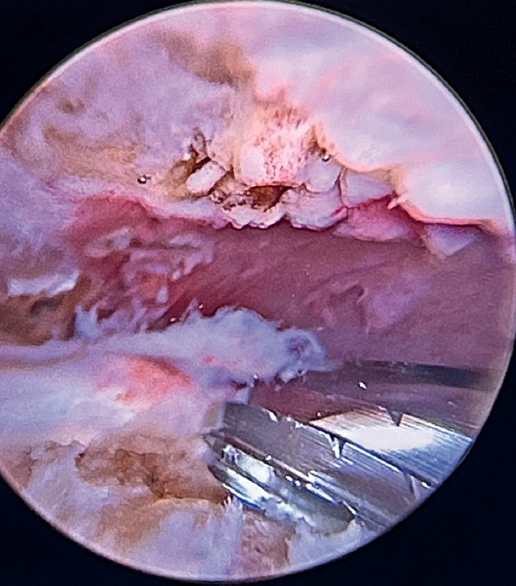
Previously, at intra-articular level, we assessed the quality of the LPB and its insertion in the supraglenoid tuberosity; from subacromial, having prepared the bony footprint on the greater tuberosity, we proceed to release the proximal portion of the tendon from the bicipital groove. The upper part of the transverse humeral ligament is sectioned to mobilize the LPB without damaging it (Figure 2). At this time, the mobility and integrity of the LPB is checked with tendon pulling forceps (Figure 3).
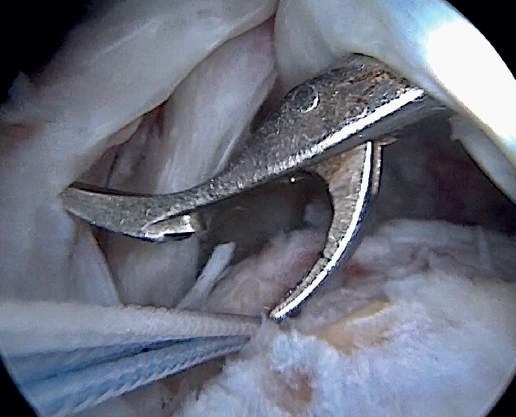
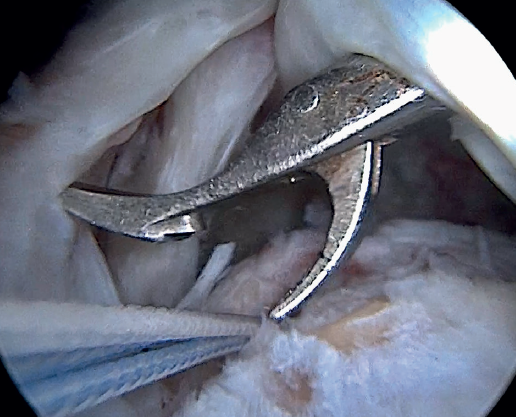
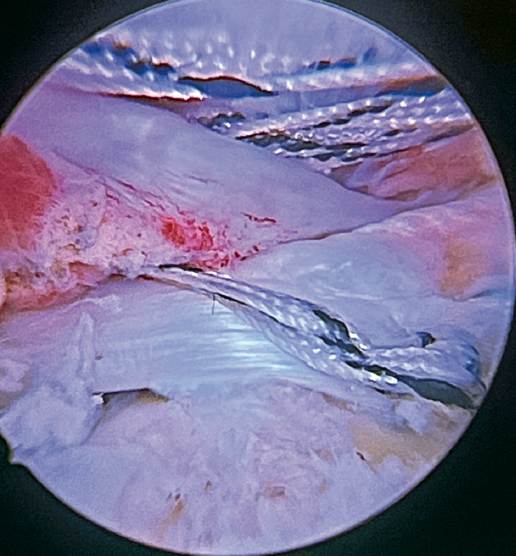
The glenoid origin of the LPB is preserved at all times. In the anterolateral zone of the greater tuberosity, we implant an anchor loaded with three sutures (FiberTak® 2.6 mm, Arthrex, Naples, FL, USA), more medial to the reflection zone of the LPB. To ensure correct guidance at implantation, we create a superolateral accessory portal without a cannula, close to the limit of the acromion.
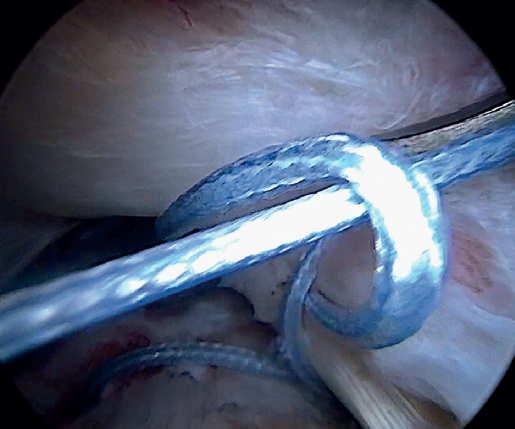
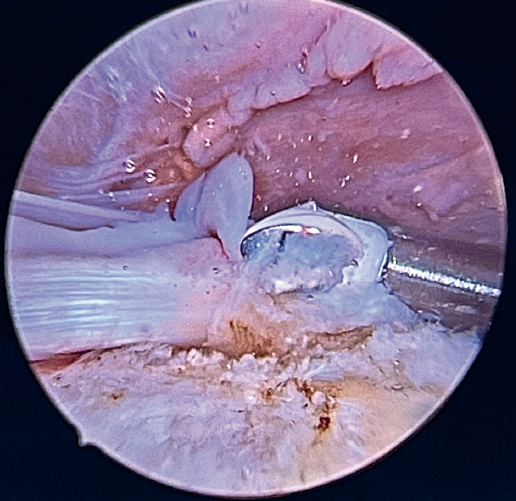
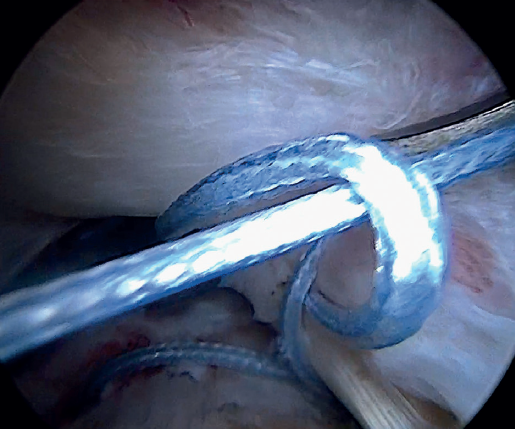
Using penetrating forceps through the anterior portal, the surgeon penetrates the LPB and retrieves one of the suture threads, performing a self-locking Lasso suture in the same step. This is done with two of the three anchoring threads to ensure a correct LPB grip to the footprint of the greater tuberosity and that the tendon does not fray; we do not cut the two threads after knotting (Figures 4, 5 and 6). We also consider it very important to apply the biceps tendon, keeping it as flat and anatomical as possible, avoiding twisting or excessive distension of the tendon. Subsequently, a tenotomy of the LPB distal to these sutures is performed (Figure 7), thus allowing the LPB to function as a superior capsular reconstruction, restricting the ascent of the humeral head.
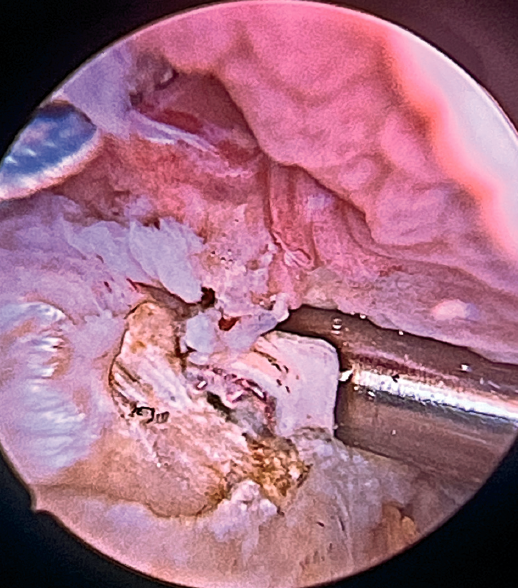
Double-row rotator cuff repair with addition of the LPB graft
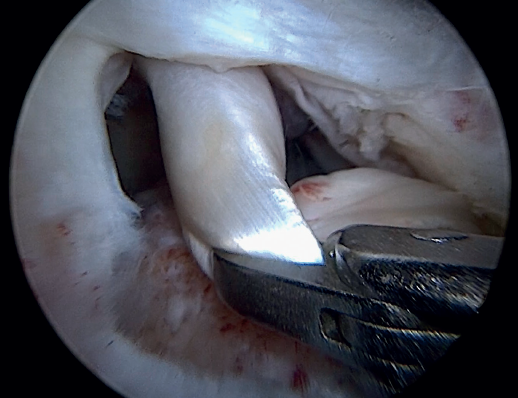
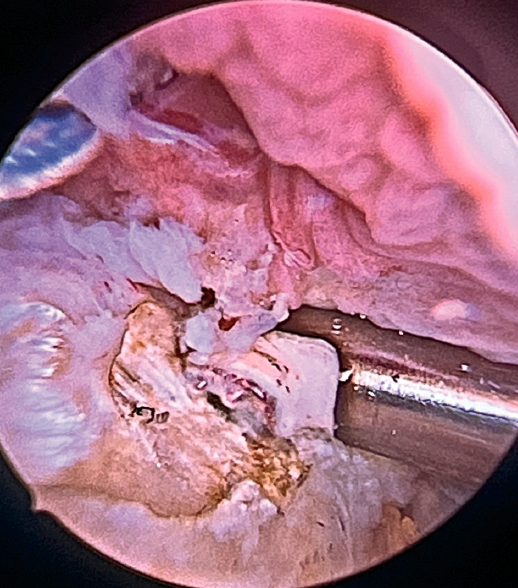
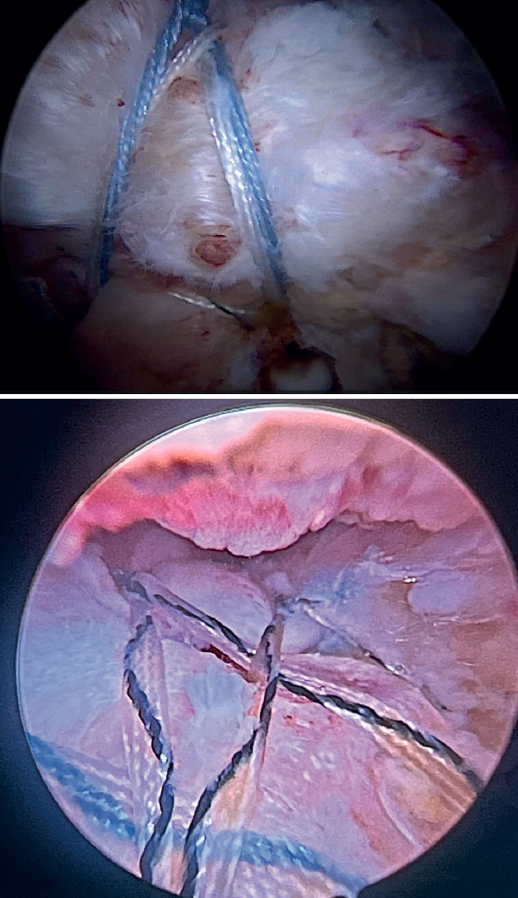
Tension-free repair of the supraspinatus tendon is then made, using the free thread left at the anchoring and taking advantage of the two sutures passed through and knotted to the proximal LPB fragment (Figures 8, 9 and 10).
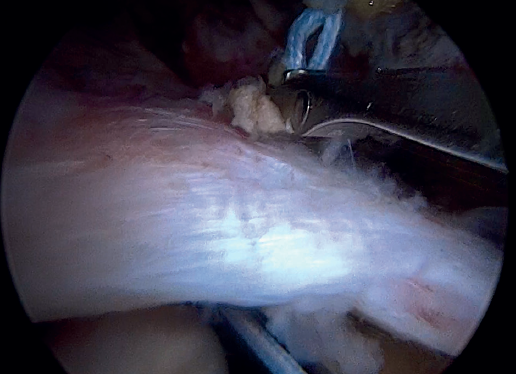
The free suture remaining at the anchoring is passed through the anterolateral supraspinatus using a Scorpion® direct suture passer (Arthrex, Naples, FL, USA), as a sliding mattress suture over the implant.
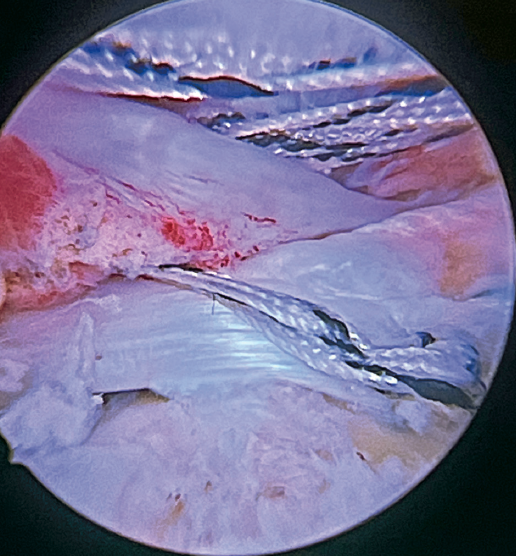
One end of each of the sutures exiting the LPB is passed through the supraspinatus likewise using a Scorpion® direct suture passer (Arthrex, Naples, FL, USA). All the threads are left unknotted and are recovered through the anterior working portal so that they do not get in the way when working later on. Note that the two threads exiting the LPB and passing through the supraspinatus will not slide; it is therefore advisable to knot them and lower them later on with a knot pusher. This point allows repair of the anterior supraspinatus to be combined with fixation of the LPB in the anterolateral zone of the greater tuberosity.

A second anchorage loaded with two tapes is implanted at the same height as the first, but in a more posterior position. Again using direct suturing with mattress stitches, we repair the posterolateral part of the supraspinatus and even the anterior part of the infraspinatus, if necessary.
Having passed both the anterior and the posterior sutures, we proceed to knot, through the lateral portal with a working cannula, each of the sutures, starting posterior and ending anterior.
Finally, the anterolateral supraspinatus threads are used in combination with the posterolateral tapes to establish a more lateral double row with two direct anchorings that allow the threads to be threaded through two loops(Biocomposite SwiveLock® C 4.75 × 19.1 mm, Arthrex, Naples, FL, USA). This achieves a tension-free double row repair of the supraspinatus, covering the superior capsular repair made with the LPB (Figure 11).
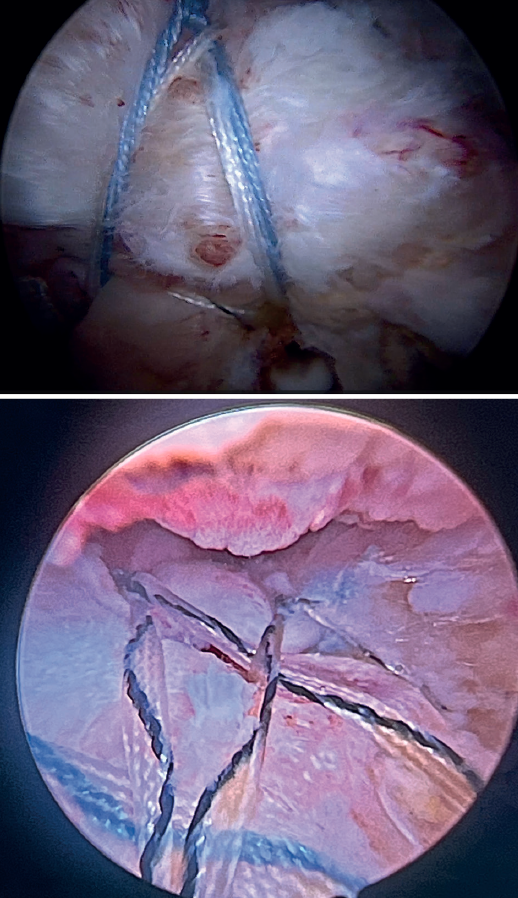
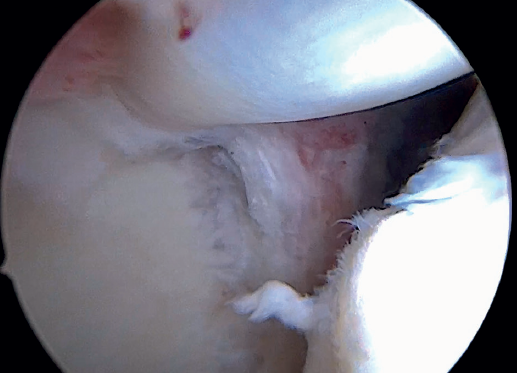
Once suturing is complete, the arthroscope can be reinserted into the glenohumeral joint to check the final result of the superior capsular reconstruction with the LPB and, especially, the correct repair of the supraspinatus to its footprint on the greater tuberosity (Figure 12).
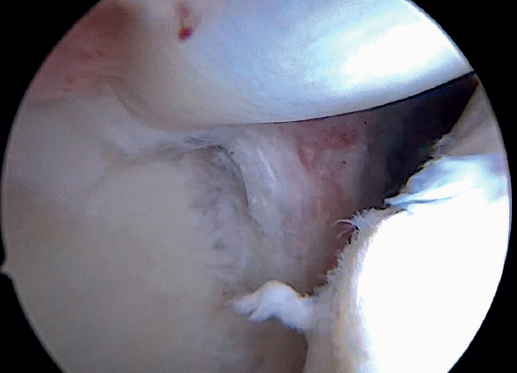
Depending on the size of the rotator cuff injury and whether it also involves the infraspinatus or subscapularis, we should be able to use more anchors to perform the repair and spread out the rotator cuff tension forces.
Closure, immediate postoperative period and rehabilitation process
At the end of surgery, the arthroscopic portals are sutured with 3-0 monofilament nylon, absorbent dressing is applied, and a sling is placed on the affected limb.
After the operation, the patient follows a standard rehabilitation protocol for rotator cuff repair. The arm is placed in a sling for 6 weeks. Active hand, wrist and elbow exercises are permitted from the outset. However, we protect the suturing by delaying passive self-assisted and pendulum exercises until the beginning of the third postoperative week. From the start of the fifth week we introduce active exercises and delay active resistance exercises until the eighth week. From the tenth week onwards there are no limitations for activities of daily living, except heavy weights (over 10 kg).
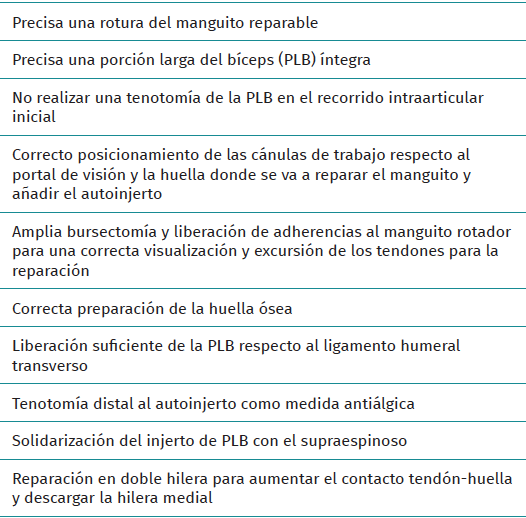
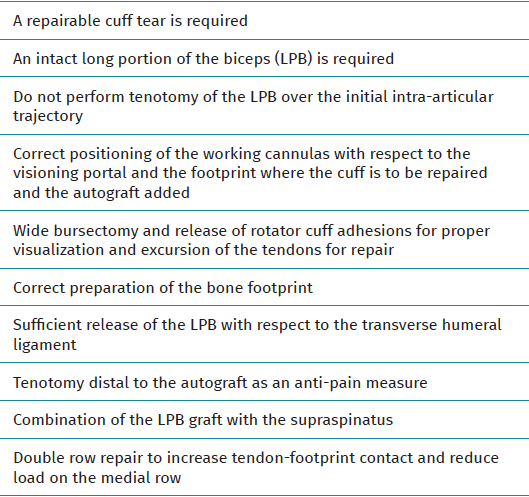
Key points of the technique (Table 1)
Discussion
Conceptually, dynamic stabilization of the humeral head, provided by the rotator cuff in elevation movements above the head, should be distinguished from the pure static stabilization provided by the superior capsule, which prevents superoanterior migration of the humeral head(17,18). The superior capsular reconstruction technique described by Mihata et al.(6) and later developed by other authors with its variants, aims to alleviate pain and disability in massive irreparable rotator cuff tears. It should be remembered that at the time Mihata described his technique, reverse total shoulder replacement was not approved in Japan. On the other hand, in repairable cuff tears, our group considers that repair of the tear, augmented by using LPB as a static stabilizer in the way of a superior capsule, guarantees a greater possibility of biological repair of the cuff, and moreover maintains correct dynamism of the glenohumeral joint. LPB plasty allows and supports tension-free, double-row cuff suturing and is cost-effective compared to traditional superior capsular reconstruction in terms of the number of total implants. This experience is also corroborated by other groups(15,19).
Superior capsular reconstruction with autologous LPB adds a strengthening factor to supraspinatus suturing, both biomechanically and biologically. Adding this superior static stabilizer probably minimizes the mid- to long-term cuff rupture rate, and seeks to improve the long-term clinical outcomes(9).
As has already been described by other authors, in this surgical technique, the fact that autologous LPB is used reduces the surgery time, is cost-effective compared to allografts or xenografts, and is cost-effective in terms of the number of total implants, as none are implanted on the glenoid side. Furthermore, the technique reduces morbidity at the autograft donor site, since the autologous LPB is in situ, and affords more preserved vascularization that may help in local healing, as the origin of the biceps in the glenoid is not altered(16,19). This technique offers a simpler and more cost-effective solution with minimal risks to the patient, stabilizing the shoulder and protecting the rotator cuff repairs.
Although the incidence of infection after shoulder arthroscopy is low overall(20), we know that it is higher in the specific case of superior capsular reconstruction(21,22). This is probably related to the increased surgery time, the use of external allografts or xenografts, and the need to introduce them through the arthroscopic portals, which implies greater implant-skin contact. These risks are also reduced with the use of autologous LPB.
We know of the scientifically proven clinical benefit of the therapeutic effect of LPB tenotomy/tenodesis in cases of tendonitis or bicipital instability, in terms of the improvement of postsurgical pain and functional outcomes(23).
In many cases, cuff tears are associated with painful bicipital disorders. This technique offers double benefit, generating superior capsule-type static stabilization with LPB and alleviating the bicipital symptoms by means of the tenotomy in most cases, and tenodesis in selected patients.
As already described by Cañete et al. in their study on this same technique(19), the LPB autograft is a capable graft, exhibiting resistance superior to that of the supraspinatus itself (32.5 vs. 16.5 MPa), with an average diameter of 6.6 mm; this is in line with the recommended sizes for the other grafts (6 to 8 mm), and duplication to reach this diameter is not required. Furthermore, in our technique, as it is performed in the anterolateral zone, ample contact space is left between the supraspinatus and the bony footprint of the greater tuberosity, increasing the chances of healing of the bone-tendon junction. It seems logical that the addition of LPB, increasing the strength of the final construction, may influence the decrease in re-rupture rate after repair. However, this is the presentation of a surgical technique, and more studies are needed to corroborate this hypothesis which, although logical, remains unproven.
We cannot end this description of the surgical technique without commenting on its limitations. The main limitation of the technique is that it requires a LPB to be present and of adequate quality and thickness, which is not always feasible in situations of chronic or extensive tears. Although postoperative pain has been described in other studies in relation to the use of LPB(9,15,23), in our experience and that of other authors(8,16,19), this is not the case. Complications inherent to LPB tenotomy therapy, such as Popeye's sign or discomfort on resisted pronation-supination, are also present here; however, they do not pose a significant medium- to long-term functional or clinical problem.
Conclusions
The use of the LPB as an in situ autograft for superior capsular reconstruction adjuvant to surgical repair of the rotator cuff is a simple, reproducible, cost-effective technique in our setting that can reduce the re-rupture rate. It therefore should be seen as a necessary routine technique in the arthroscopic shoulder surgeon's surgical armamentarium.
Supplementary material
The video of the technique accompanying this article can be seen at (Video 1):